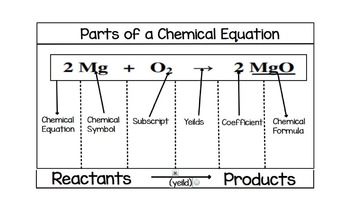
45 labels of a chemical equation
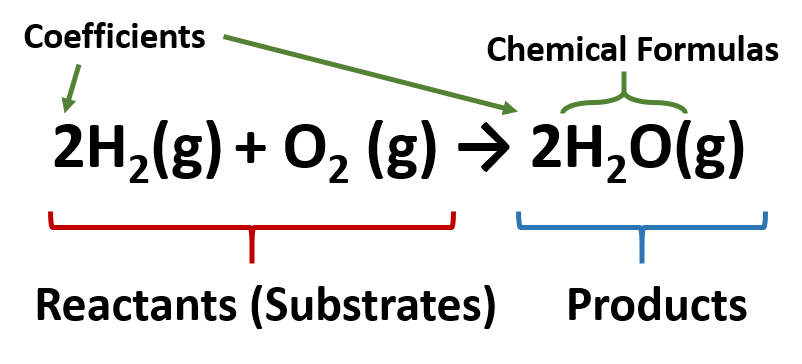
Chemical Equations - Let's Talk Science A chemical equation is a way to represent a chemical reaction. The reactants are given on the left hand side and the products are given on the right hand side. Instead of an equal sign, an arrow is drawn between the reactants and the products. It is important to note that the arrow does not mean the same thing as an equal sign. Examples of Balanced Chemical Equations - ThoughtCo A balanced equation contains the same number of each type of atoms on both the left and right sides of the reaction arrow. To write a balanced equation, the reactants go on the left side of the arrow, while the products go on the right side of the arrow. Coefficients (number in front of a chemical formula) indicate moles of a compound.
Symbols in Chemical Equations - Harper College Symbols in Chemical Equations. Symbol: Meaning + used to separate one reactant or product from another used to separate the reactants from the products - it is pronounced "yields" or "produces" when the equation is read used when the reaction can proceed in both directions - this is called an equilibrium arrow and will be used later in the ...

Labels of a chemical equation
Symbols used in Chemical Equations Flashcards | Quizlet used for reversible reactions. +. used to separate two reactants or two products. ∆ (over the reaction arrow), indicates the application of heat to the reactants. ↓. indicates a precipitate formed by a reaction. (aq) designates an aqueous solution; the substance is dissolved H₂O; placed after the chemical formula. Chemical Ingredients 101: How to Read a Product Label All labels are required to have the following: Pictograms A signal word Hazard and precautionary statement The product identifier Supplier identification OSHA's Hazard Communication Standard pictogram provides more information about how to read these labels. 10 Sources 1 Toothpastes | American Dental Association (ada.org) Chemical Symbols and Formulas - SchoolWorkHelper No numbers are required in this formula because only one atom of each element is involved in making salt. - If more than one atom of an element is involved in a chemical, a number must be included in the chemical formula. The numbers in chemical formulas are called subscripts. Subscripts are small numbers written below the letters. - For ...
Labels of a chemical equation. Balancing Chemical Equations Activity - Carolina Knowledge Center It may be helpful to crease a sheet of notebook paper in half and label the left side "reactants" and the right side "products." ... The coefficient is the number in front of the chemical formula in a chemical equation, indicating the number of molecules. (Absence of a coefficient is understood to indicate 1 molecule.) How do you label a chemical equations? - Answers What are chemical equations? chemical equations are a formal algebra that permits the calculation of chemical reactions. What is a Chemical Equation? - Definition & Examples A chemical equation provides information about the correct proportions of ingredients that are needed to make new substances in chemical reactions. To make a large glass of lemonade, use 1 1/2 ... Chemical formulae - LaTeX Cookbook Chemical formulae and equations have a different style compared to mathematical formulae and equations. For example: Letters mean atomic symbols and are written upright, unlike italic math variables. Numbers are commonly used in subscripts, indicating the number of atoms. We use a lot of subscripts and superscripts, they should be aligned properly.
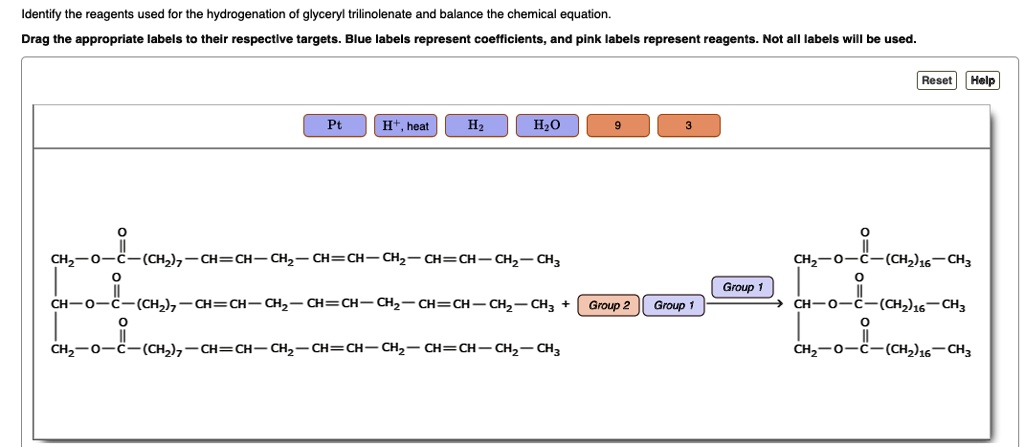
What Are Chemical Equations? - ThoughtCo This can be seen in the following equation: 2 H 2 (g) + O 2 (g) → 2 H 2 O (l) Hydrogen and oxygen are indicated by (g), which means they are gases. Water is marked (l), which means it is a liquid. Another symbol you may see is (aq), which means the chemical species is in water — or an aqueous solution. The (aq) symbol is a sort of shorthand ... Label the chemical equation. 2 Mg(s) + O2(g) → 2 MgO(s ... - Brainly.com Click here 👆 to get an answer to your question ️ Label the chemical equation. 2 Mg(s) + O2(g) → 2 MgO(s) Label the chemical equation. 2 Mg(s) + O2(g) → 2 MgO(… lucy5221 lucy5221 07/23/2020 Chemistry High School answered • expert verified Label the chemical equation. Solved Listed are examples of several types of chemical | Chegg.com Chemistry. Chemistry questions and answers. Listed are examples of several types of chemical equations. Note that a combination reaction and a synthesis reaction are the same type of reaction. Each reaction is ony partially balanced Complete the task of balancing each chemical equation Drag the appropriate labels to their respective targets. How to Read a Chemical Label | Understanding Chemical Labels There can be none, one, or more than one Pictogram on a label, depending on the nature of the chemical. 10. More technical in nature, also shown on our labels is your "Right to Know." It lists the top 5 chemical ingredients that are in a formula, based on percentages. Almost all of our chemical formulations will list water as the first ingredient.
Balanced chemical equation - BYJUS (Oxygen = 4). Then we multiply the number of oxygen atoms on reactant side by 4, such that the number of oxygen atoms on both sides of the reaction is balanced. The equation becomes Now the number of hydrogen atoms is 8 on the reactant side which is more than that of the product side, hence we need to balance hydrogen atoms, and hence we multiply How do you Write a Chemical Equation? - A Plus Topper The products of reaction are written in terms of their symbols or molecular formulae on the right-hand side of the equation. A plus (+) sign is added between the formulae of the products. In between the reactants and the products an arrow sign ( ) is inserted to show which way the reaction is occurring. A + B C + D Table of Content - BYJUS In order to describe a state of chemical equilibrium, the symbol '⇌' is used. To denote stoichiometric relationships, the '=' symbol is used. In order to describe a reaction that occurs in both forward and backward directions, the symbol '⇄' is used. Chemical Nomenclature and Chemical Formulas - Owlcation There are 2 atoms of Hydrogen and 1 atom of oxygen The formula of glucose is C6H12O6 There are 6 atoms of Carbon, 12 atoms of Hydrogen and 6 atoms of Oxygen. In writing formulas, the total positive charges plus the total negative charges must equal to zero since the compound is electrically neutral. Examples of common, simple and polyatomic ions
Writing and Balancing Chemical Equations - Chemistry 2e The physical states of reactants and products in chemical equations very often are indicated with a parenthetical abbreviation following the formulas. Common abbreviations include s for solids, l for liquids, g for gases, and aq for substances dissolved in water ( aqueous solutions, as introduced in the preceding chapter).
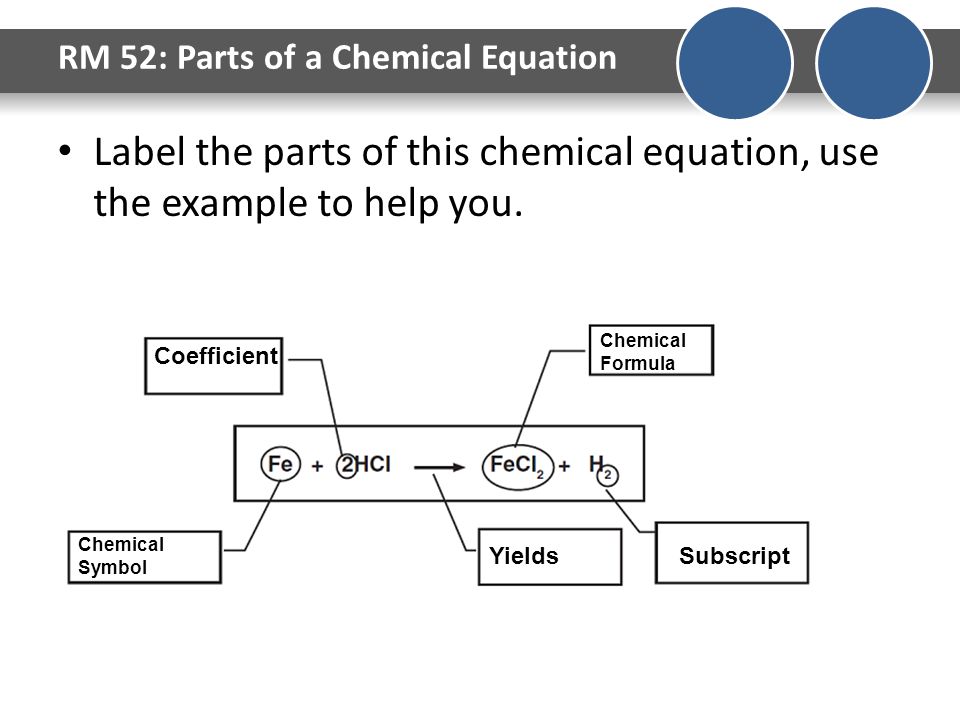
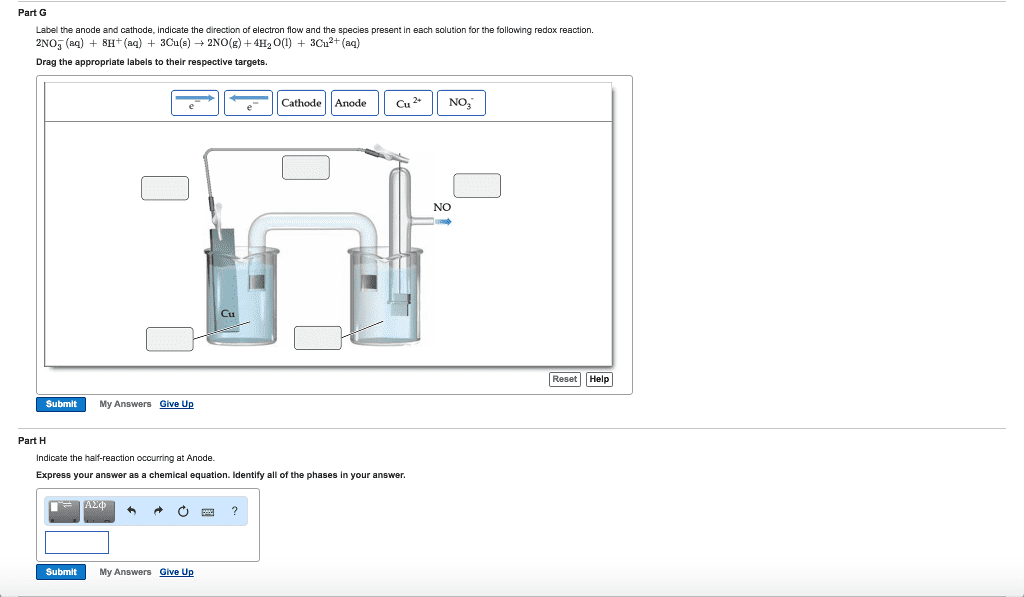
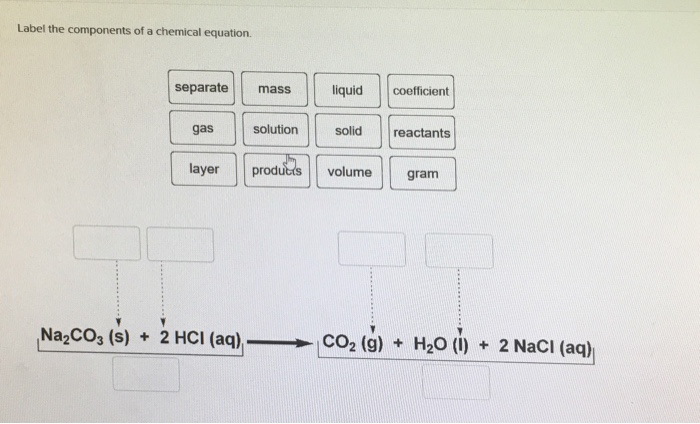
Solved Label the components of a chemical equation. separate | Chegg.com Chemistry Chemistry questions and answers Label the components of a chemical equation. separate mass liquid coefficient gas solution solid reactants layer prod uiets volume gram Na2co, (s) + 2HCl (aq),- ?.co2 (g) + H2O (l) Co2 (g) + H20 ) + 2 Naci (aq)
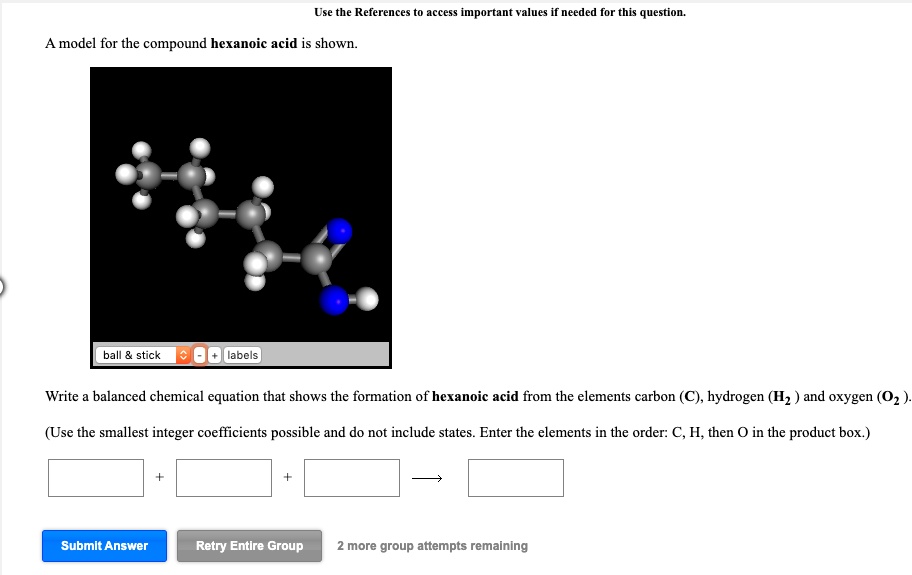
The Chemical Equation - Introductory Chemistry - 1st Canadian Edition Start by writing the chemical equation in terms of the substances involved: C 2 H 6 + O 2 → CO 2 + H 2 O We have two carbon atoms on the left, so we need two carbon dioxide molecules on the product side, so that each side has two carbon atoms; that element is balanced.
4.1 Writing and Balancing Chemical Equations | Chemistry The physical states of reactants and products in chemical equations very often are indicated with a parenthetical abbreviation following the formulas. Common abbreviations include s for solids, l for liquids, g for gases, and aq for substances dissolved in water ( aqueous solutions, as introduced in the preceding chapter).
How would you label each formula in the chemical equation below as ... Explanation: The reactants are on the LEFT HAND SIDE of the equation. The products are on the right hand side. So for the oxidation of iron, F e +S → F eS Iron and sulfur are the reactants, and iron sulfide is the product. Answer link
Chemical equation - Wikipedia a chemical equation is the symbolic representation of a chemical reaction in the form of symbols and formulae, wherein the reactant entities are given on the left-hand side and the product entities on the right-hand side with a plus sign between the entities in both the reactants and the products and an arrow that points towards the products, and …
How to Write a Chemical Equation (with Pictures) - wikiHow All chemical equations look something like "A + B →C (+ D...)," in which each letter variable is an element or a molecule (a collection of atoms held together by chemical bonds). The arrow represents the reaction or change taking place.
Writing and Balancing Chemical Equations - Course Hero Balancing Equations A balanced chemical is equation has equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides.This is a requirement the equation must satisfy to be consistent with the law of conservation of matter. It may be confirmed by simply summing the numbers of atoms on either side of the arrow and comparing these sums to ...
5.2 Chemical Equations - Lumen Learning It is not uncommon to include a phase label with each formula— (s) for solid, (ℓ) for liquid, (g) for gas, and (aq) for a substance dissolved in water, also known as an aqueous solution. If we included phase labels for the reactants and products, under normal environmental conditions, the reaction would be as follows: H2(g) + O2(g) → H2O (ℓ) Note
What are the Parts of a Chemical Equation? - Life Persona The steps followed to write a chemical equation are: - Reagents and reaction products are identified and noted. - The formula or symbols of the reagents are written on the left side with a '+' sign between them. - The formula (s) of the products are written on the right side with a '+' sign between them.
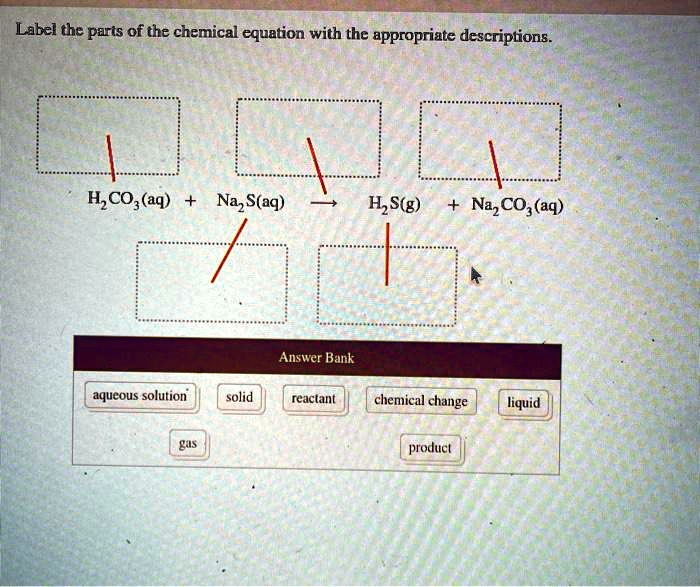
Parts of a chemical Equation- Write an equation and label the following ... Parts of a chemical Equation- Write an equation and label the following parts a. Coefficient, subscript b. Atoms, molecules C. Reactants and products Expert-verified answer mapooja789 Coefficient, subscript, Atoms, molecules, Reactants, and products are parts of a balanced chemical equation Balanced chemical equation 6CO₂ ₊ 6H₂O=1C₆H₁₂O₆₊61O₂
Chemical Symbols and Formulas - SchoolWorkHelper No numbers are required in this formula because only one atom of each element is involved in making salt. - If more than one atom of an element is involved in a chemical, a number must be included in the chemical formula. The numbers in chemical formulas are called subscripts. Subscripts are small numbers written below the letters. - For ...
Chemical Ingredients 101: How to Read a Product Label All labels are required to have the following: Pictograms A signal word Hazard and precautionary statement The product identifier Supplier identification OSHA's Hazard Communication Standard pictogram provides more information about how to read these labels. 10 Sources 1 Toothpastes | American Dental Association (ada.org)
Symbols used in Chemical Equations Flashcards | Quizlet used for reversible reactions. +. used to separate two reactants or two products. ∆ (over the reaction arrow), indicates the application of heat to the reactants. ↓. indicates a precipitate formed by a reaction. (aq) designates an aqueous solution; the substance is dissolved H₂O; placed after the chemical formula.





























Post a Comment for "45 labels of a chemical equation"